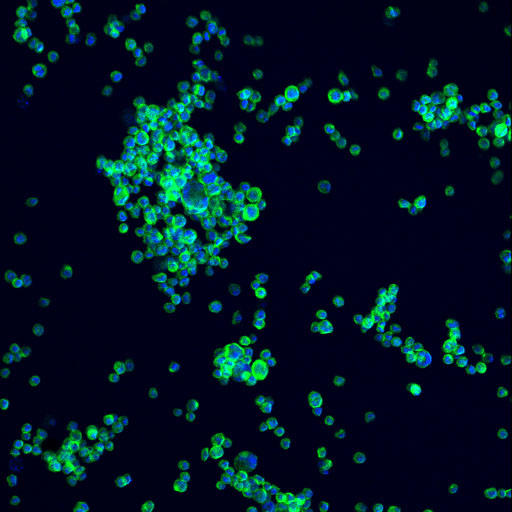
NIST has developed two new reference materials to enhance quality control in the biopharmaceutical industry. One is a living reference material, NISTCHO, which produces monoclonal antibodies and allows manufacturers to optimize their production processes. The other is a standard reference material, SRM 1989, consisting of precisely sized particles designed to help detect and quantify impurities in protein-based drugs. These materials aim to accelerate drug development, ensure drug safety and efficacy, and reduce manufacturing costs by improving the accuracy and uniformity of quality control measures. AI
Summary written by gemini-2.5-flash-lite from 2 sources. How we write summaries →
RANK_REASON NIST, a government research agency, released new standard reference materials for the biopharmaceutical industry.